Outcomes, Outputs and Progress
Serine-type site-specific DNA recombinases are valuable tools for conditional expression of genetic constructs. We have a particular interest in mammalian neurobiology, where the currently small number of efficient serine recombinases limits the role these enzymes can play in elucidating (for example) neural connectivity. The core aim of this project is to identify and optimise serine recombinases for function in mammalian cells.
We have 1) constructed plasmid-based fluorescence reporters assays for recombinase activity, 2) demonstrated the function of these reporter assays in human fibroblast cells, 3) designed codon-optimised open-reading frames for a set of 6 recombinases whose functions have not previously been tested in mammalian cell lines. Having validated our reporter system, we plan to have these codon-optimised genes synthesised and to insert them into our validated vector backbones for characterisation.
Genetic constructs
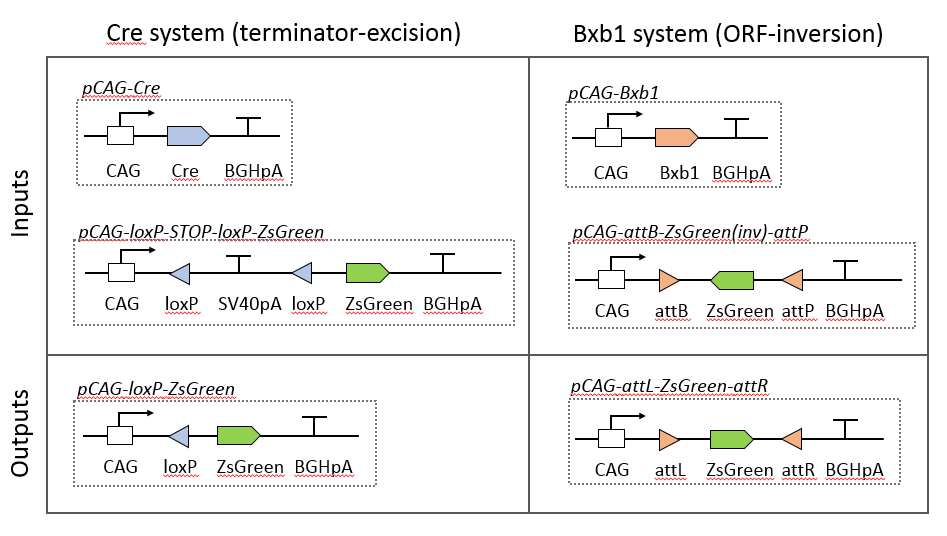
Figure 1. Design of recombinase reporter and expression systems. Co-transfection of a recombinase expression plasmid (e.g. pCAG-Cre) with its cognate reporter plasmid (e.g. pCAG-loxP-STOP-loxP-ZsGreen) should result in recombination of the reporter and hence expression of ZsGreen fluorescent protein.
In order to assess recombinase efficiency we tested two designs (see figure 1):
1) A terminator-excision system consisting of a transcriptional termination region flanked by recombinase recognition sites: removal of the transcriptional terminator facilitates RNA polymerase progression through the reporter ORF.
2) An ORF-inversion system consisting of an inverted reporter ORF (ZsGreen) flanked by recombinase recognition sites.
We have assembled and tested a terminator-excision system for Cre (a tyrosine recombinase) and an ORF-inversion system for Bxb1 (a serine recombinase system). We chose Cre and Bxb1 for our test systems because these recombinases are among the most efficient DNA recombinases for excision and inversion reactions, respectively. We are using these systems to validate our strategy and will use them as references for the activities of other recombinases (see below). We in the process of constructing a terminator-excision system for Bxb1 for comparison.
Validation of reporter constructs
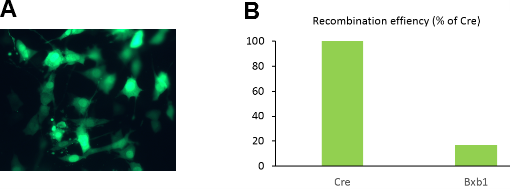
Figure 2. Validation of reporter and recombinase expression constructs. A) Co-transfection of Cre reporter (pCAG-loxP-STOP-loxP-ZsGreen) and recombinase constructs (pCAG-Cre). Green cells indicate successful recombination events. B) Relative recombination efficiencies of Cre and Dre reporter/expression systems.
The above plasmids were transfected into NIH 3T3 human fibroblasts to test for recombinase activity. Images of cells were captured using epifluorescence microscopy at 24 hours post-transfection. Transfection with reporter plasmids alone resulted in low background fluorescence that could easily be distinguished from constitutively fluorescent cells. As hoped, co-transfection of recombinase and reporter plasmids resulted in populations of brightly fluorescence cells, illustrated in figure 2A. Green cells in figure 2A indicate successful recombination of plasmid pCAG-loxP-STOP-loxP-ZsGreen by Cre recombinase expressed from plasmid pCAG-Cre. Similar results were obtained for the Bxb1 ORF-inversion system, though with lower efficiency (see figure 2B).
Ongoing work
- Characterize the reporter constructs using flow cytometry.
- Construct optimised recombinase expression cassettes.
- Characterize the full set of expression cassettes by microscopy and flow cytometry.
- Share reporter and recombinase plasmids via Addgene as well as associated online methods for open access to the scientific community.

